 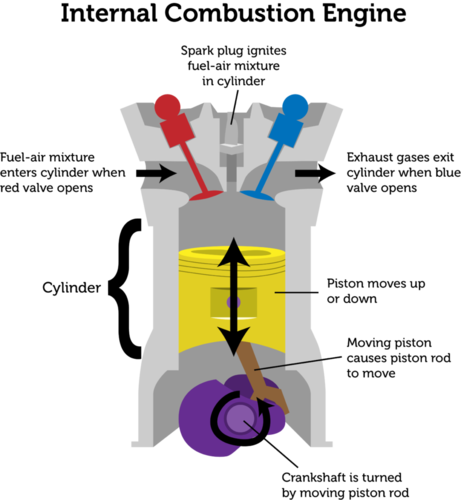
Process heating equipment are rarely run that way. When burned all fuel and air is consumed without any excess left over. The stoichiometric ratio is the perfect ideal fuel ratio where the chemical mixing proportion is correct. 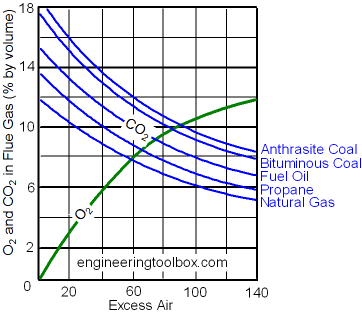 
To determine the excess air or excess fuel for a combustion system we starts with the stoichiometric air -fuel ratio. With unburned components in the exhaust gas such as C, H 2, CO, the combustion process is uncompleted and not stoichiometric. Stoichiometric or Theoretical Combustion is the ideal combustion process where fuel is burned completely.Ī complete combustion is a process burning all the carbon (C) to (CO 2), all the hydrogen (H) to (H 2O) and all the sulphur (S) to (SO 2). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
